Different types of relation of partition function with different parameters:
a) Relation with internal energy of the system: The partition functions are related to the Thermo-dynamical parameters.

It is clear from the above equation that internal energy of the system is proportional to that of partition function. i.e. the amount of vibrational, rotational and translational energy changes are directly related to the internal energy of the medicine.
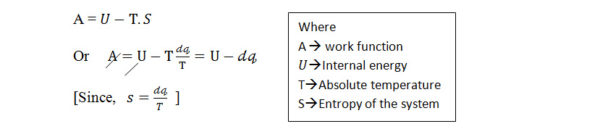
b) Relation of internal energy with Thermo-dynamic work function:
As we know, from the Thermo-dynamic work function,
c) Relation of free energy with partition function:
We know that free energy is related to partition function:
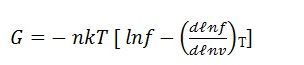
Relevancy of viscosity: Here viscous force or viscosity will be neglected although a molecular collision among the drug and alcohol molecules occurs during drug-dynamization.
We know that a viscose force is a Tangential Force achieved due to friction between the two layers of the fluid in a particular direction and when the flow is at steady state. But during drug-dynamization, molecular collisions occur haphazardly and steadiness is lost. So, the viscous force is meaningless here.
Calculation of Electromagnetic energy: Electro-magnetic energy change will occur only when the drug molecules are in polar character with smaller molecular radii e.g. Hydrofluoric acid (HF). Here during molecular rotation, the molecules achieve a dipole movement as electro-magnetic energy change will occur that will be absorbed by ethyl alcohol molecules and stability will be formed via solvent molecule interaction.
During drug-dynamization, the rotational changes among the drug and vehicle molecules may occur due to ten equal strokes. During rotational motions, some drug molecules which are polar in character produce an electro-magnetic field, e.g., Fluoric acid (HF), Nitric acid (HNO3) etc.
So, electro-magnetic energy may be formed during rotational changes which can be qualitatively measured in the following way by means of Quantum Mechanics:
We know that

Calculation of Resonance Energy: If the drug molecules have a delocalized system and the medium (ethyl alcohol) becomes polar in character, then resonating energy changes may occur.
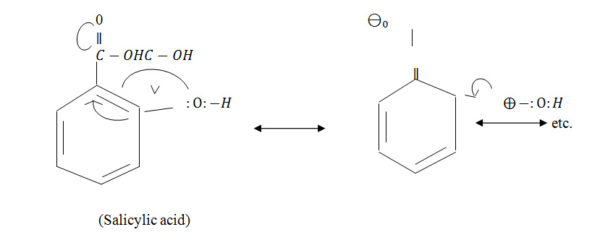
During preparation of the medicines, some drugs that possess different resonating structures are used. e.g., salicylic acid etc. During dynamisation of the drug, some energy will be required for the resonating change of the structure and in order to acquire the stability, some energy will be emitted in the solution of the vehicle molecules. And, this energy is called the Resonance Energy; which will be collected by the vehicle molecules and stored as Internal energy of the medicinal solution.
Or the size of the drug molecules become comparatively high, then due to steric hindrance rotational energy changes do not take place.
If rotational energy changes occur, then we can establish the expression of the rotational energy changes as follows:
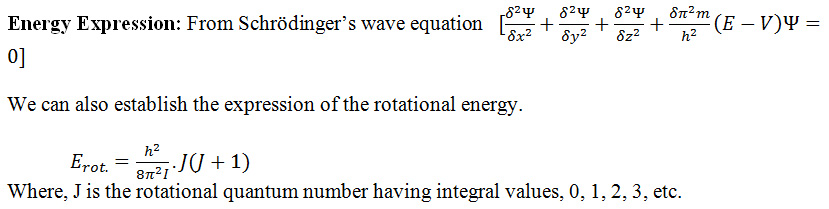
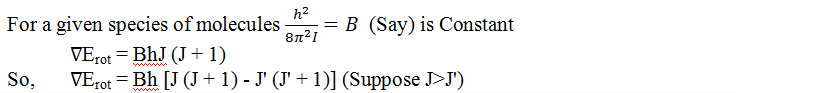
From this equation we can calculate the value of rotational energy change of a diatomic molecule.
Higher the number of resonating structures, higher will be the resonating energy and higher will be the stability and the released energy will be high which helps to grow up the Internal energy of the medicinal solution.
Calculation of the Resonance Energy: The dependence of the energy of resonance between two structures is discussed below:-

V→ Potential energy. The integral is taken over the whole of configuration space for the system, is the complex conjugate of This equation helps to calculate the Resonance Energy of the medicinal molecules.
And in this way, different types of energy are produced during drug dynamisation;
i. Vibrational energy,
ii. Rotational energy,
iii.Transational energy,
iv. Electromagnetic energy,
v. Resonance energy, etc.
These energies are absorbed in alcohol and lactose sugar as internal energy in the molecular bond and also H-bonding network structure of ethyl alcohol and lactose sugar via solvent molecules interactions. Since the amount of total energy produced by drug dynamisation is minute in nature, so it cannot be measured experimentally and the energy will be qualitative in nature. Calculation of energy absorbed in the medicinal vehicle.
Energy absorbing power of:
- Ethyl alcohol
- Lactose sugar (milk sugar)
From the structural point of view, it is clear that ethyl alcohol forms tetrahedral structure where 2 electrons of 2P orbital take part in forming Co-valiant bond with H-atom and Carbon atom of ethyl group. So two electrons (lone pair) exist in the 2S orbital and〖2P〗_x sub level. Therefore, it forms 〖SP〗^3- hybridization. The structure is as follows:

C_2 H_5 OH molecule via H-bonding due to electrostatic force of attraction Structure of and therefore stability of becomes high and acts as a good solvent. Now, due to drug –dynamization (succussion with ten equal strokes) a minute amount of energy is emitted by the drug and vehicle substances which can be easily taken up by ethyl alcohol molecule.
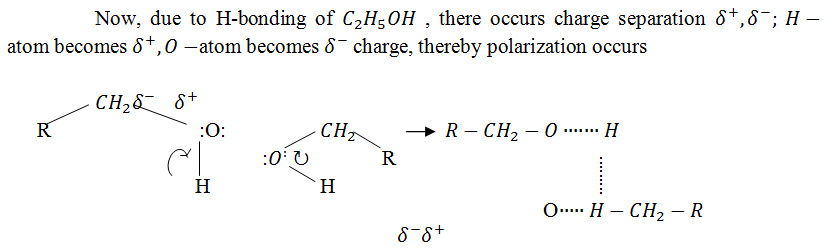
Now, electrons of –O-H bond shift towards the O-atom due to higher electro-negativity of oxygen atom than that of H-atom and thereby H-atom becomes deficient in electron and it attracts the –ve end of another O-atom of other C_2 H_5 OH molecule and so there exists a great probability of electron to shift the lone pair of electron from one molecule toward the other due to electrostatic force of attraction. Now, the emitted energy by drug-dynamization is absorbed by ethyl alcohol and lone pair of electron shift towards the H-atom to form a stable structure of C_2 H_5 OH. Since, the amount of emitted energy is minute in nature, So, this energy is easily absorbed by C_2 H_5 OH molecule and it cannot affect on the stability of the alcohol. This energy is stored as bond energy of the molecular bond of C_2 H_5 OH molecule. This energy may be calculated by measurement of bond energy of medicinal alcohol and bond energy of the pure C_2 H_5 OH. From the differentiation, we can evaluate the amount of the emitted energy.
Role of milk sugar for the preparation of medicines:-
But, in the case of preparations of some modern medicines, 1st product (mother) is produced by the absorption of radiation by milk sugar (lactose) in saturated condition (e.g., X-ray, Electricity, Elactrictus, Magnetic Pole Ambo etc).The se radiations are of higher frequencies. So, the molecular bond of C_2 H_5 OH can be dissociated by absorption of such radiations. So, these will be absorbed in milk sugar (lactose) as it has more stability than that of C2 H_5 OH. The structure of milk suger is as follows:-
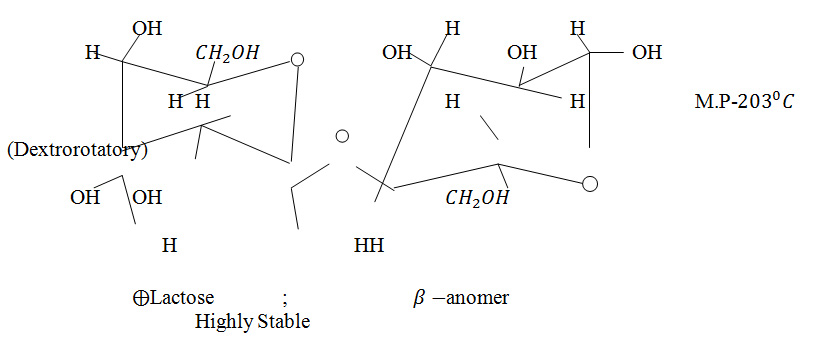
Now, to diminish the energy concentration higher potency is produced by alcoholic dilution.
The causation of higher stability of lactose sugar:-
Due to inter and intra-molecular H-bonding in lactose sugar, it attains higher stability .It contains a large no of –OH groups .So; there is a great scope to form the inter- and intra- H-bonding in lactose sugar. So, the stability becomes higher than that of ethyl alcohol.
Thermo dynamical concept: From the Thermo dynamical concept we can measure qualitatively the amount of Internal energy.
We know that during drug dynamization, there occurs a definite number of successive strokes (ten number of equal strokes) and thereby a definite amount of mechanical energy is required and some frictional forces are produced due to molecular frictions among the drug vs drug molecules , drug vs vehicle molecules , vehicles vs vehicles molecules , vehicle vs wall of the Container , drug vs wall of the Container. Now, according to the law of conservation of energy, this frictional energy will be converted into internal energy which will be absorbed in the alcohol molecules (i.e. Medicinal molecules).
From the mathematical formula we can derive

This means higher the dilution, higher will be the free energy change & internal energy change will also be high.
Causation of the specificity of the drug: We know that different drugs possess different atomic or molecular structures. So, frequency and wave length of the released energy during drug dynamization will also be different. This individual frequency produced at the very beginning of the drug dynamization process remains constant in the alcohol/ lactose matrix. This leads to the specificity of a drug.
Mechanism of action of the energised medicine in the living organism: Now, we will have to express how this energised medicine acts in the living organism to cure the patient. The frequency of the energised medicine is similar to the energy lost from the living body which is to be administered to make up the total energy (vital force). The energy stored in alcohol or lactose is absorbed via the nervous system to produce an electrical response in nerves. ln a word, the absorption of medicine in the living body maintains the electrical phenomena as all the physiological works of the living body are governed by the electro physiological phenomena.
Conclusion: We can say that a minute amount of energy absorbed in the medicinal alcohol acts as stimuli. When this medicine touches the nerves, then the receptors collects the medicinal energy (physical energy) which is transmitted via ionic conduction (Na+, K+) through the nerve fibers. Thus, the receptors behave as a biological transducer. Because, the receptors take up the physical energy and transform this energy into the electrical energy. Thus, electrical energy (electromotive force) is produced due to absorption of the medicinal energy. This energy helps to turn back the diminished total energy (Life guiding energy) to the original state (normal energy condition) and start the original bio-chemical reactions in the living body. ln a word, the medicine maintains the electro – physiological phenomena in the living organism.
References:
- Hahnemann S. Organon of Medicine. 5th & 6th ed, Dudgeon RE, Boericke, W (trans.), New Delhi: B Jain Publishers; 2011
- Kent J T. Homoeopathic Philosophy. New Delhi: B Jain Publishers; 2004
- Allen HC. Keynotes and characteristics with comparisons of some of the leading remedies of the Materia Medica, 4th Indian ed.: Kolkata: Roy Publishing House; 1977
- Rakshit PC. Physical Chemistry. Revised ed & enlarged ed edition: Sarat Book House;2014
- Pauling Linus. The Nature of the Chemical Bond and the Structure of Molecules and Crystals. Cornell University Press, 1960
- Finar IL. Organic Chemistry. Pearson Education India; 6 edition.